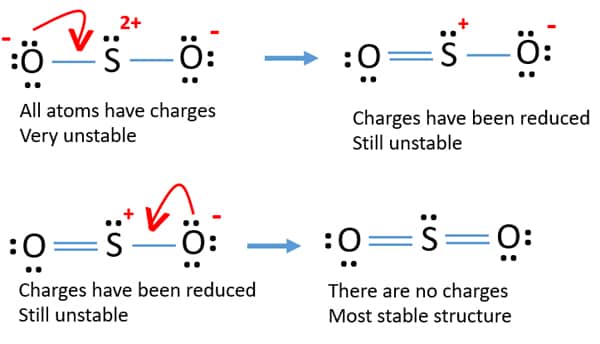
SO2 is manufactured in industrial area by burning sulfur and its components in the presence of oxygen. Scientists also have proved, that the SO2 is found in Venus and in other bodies of solar system also. It is the measure component of green house effect. There are total five lone pairs on SO2 atom. The lone pairs of oxygen atom in each of SO2 lewis structure is two. In the best reasonable structure ,the lone pairs of sulfur atom is one. This is the most reasonable structure for SO2. similarly other oxygen also has 0 formal charge. Oxygen contain 6 valence electron,2 non bonding electrons and 2 bonds So we get 6-4-2=0. This bonding electrons divided by 2, we get 4. Sulfur has 6 valence electrons, 2 non bonding electrons and 8 bonding electrons. Take 2 electrons from another oxygen to sulfur make double bond in sulfur and oxygen. The formal charge of another oxygen will be 6-4-2= 0. One of the oxygen having formal charge +1. So the Formal charge of sulfur is 6-2-3 =+1. Six bonding electrons divided by 2, we get 3 electrons. sulfur has six valence electrons, 2 non bonding and 6 bonding electrons. SO2 lewis structure of total valence electrons 18.Sulfur and oxygen has six electrons. Image credit : SO2 by istock SO2 lewis structure formal charges: The three pairs of bonding electron is in 109° angel. 
In the second way, the geometry of SO2 is trigonal planner. That is by the valence shell electron pair repulsion theory that is VSEPR theory. Where the oxygen is move away from each other, give the molecule in bent shape. 
The lewis structure doesn’t tell us directly but if we visual the atoms in unbonded electron of central atom, repelling each other. 
This is because electron are repel to each other. From 1st lewis structure of SO2, where the unbounded electrons are spread up in three dimensions. We can found the molecular geometry of SO2 in 2 ways. The smell is very much similar to the burnt matchsticks Sulfur dioxide released while volcanic eruptions. The chemical formula of sulfur dioxide is SO2. SO2 lewis structure consists of Sulphur and oxygen. But practically this is not really found. This is best lewis structure theoretically. Similarly if another electron from other oxygen bonded with sulfur than double bond will created. In this way ,sulfur and oxygen both obey the octet rule. If one electron pair of oxygen is bonded with sulfur than another lewis structure is found. This bonded with single bond than sulfur didn’t obey the octet rule. There are 6 valence electrons of sulfur and oxygen. Sulfur dioxide lewis structure of SO2, first we require the total valence electrons of both sulfur and oxygen. So we are going to study how the best structure we can determined by knowing shape, hybridization etc. We can understand the boding between atoms in a SO2 molecule. There are so many facts about the internal structure obtained from drawing of lewis structure.īy using the lewis concept we can draw the best resonate structure for sulfur dioxide. As a preservative, it protects the color of the fruit and prevents rotting.SO2 Lewis structure shows various chemical properties of the compound. It is commonly used to produce sulfuric acid, but is also used as a preservative for dried fruits because of its antimicrobial properties. Sulfur dioxide is formed by the combustion of the pure sulfur element. Thus, sp3 hybridized molecules have longer and weaker bonds than those of sp or sp2 hybridized molecules. The closer the electrons are to the nucleus, the stronger the bond. The greater the s-character, the closer the electrons are to the atom’s nucleus. Each sp3 hybrid orbital also has 25 percent s-character and 75 percent p-character. Sp3 hybrid orbitals are delocalized, meaning that each orbital occupies the same energy level. For an atom to be sp3 hybridized, it must have one s orbital and three p orbitals. In this shape, electron repulsion is minimized. Sp3 hybridization explains the tetrahedral structure of molecules with bond angles of 109.5 degrees. Hybridization of molecules also forms more stable orbitals and stronger bonds at lower energy than unhybridized compounds. Hybridization explains the molecular structure of a compound.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
